Mécanismes fondamentaux de la dissémination métastatique
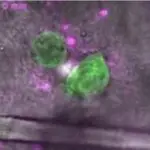
La dissémination métastatique des cellules tumorales est une étape cruciale de la progression du cancer, ayant un impact majeur sur le traitement et la survie des patients. À ce jour, il n'existe aucun traitement efficace ciblant spécifiquement la propagation métastatique des cellules tumorales. Afin de mieux comprendre les mécanismes moléculaires et cellulaires de la dissémination tumorale, tout en évitant les biais inhérents aux modèles expérimentaux, nos études reposent sur des explants tumoraux obtenus directement lors d'interventions chirurgicales réalisées chez un grand nombre de patients (« du chevet au laboratoire »).
Notre objectif est de répondre aux questions suivantes:
✓ Comment les cellules cancéreuses acquièrent-elles leurs propriétés migratoires ?
✓ Utilisent-ils une ou plusieurs stratégies pour se déplacer ?
✓ Est-il possible d'arrêter les mécanismes responsables de leur propagation ?
Trois exigences minimales pour la CAM ont été déterminées :
- Les amas cellulaires doivent polariser un réseau supracellulaire d'actomyosine qui définit l'arrière;
- Les amas cellulaires doivent se comporter comme un solide dans lequel les cellules tremblent légèrement, c'est-à-dire oscillent autour d'une position fixe au sein du groupe;
- Les tremblements doivent être suffisamment forts pour générer des forces de frottement sur le substrat pour autopropulser l'agrégat dans la direction opposée.
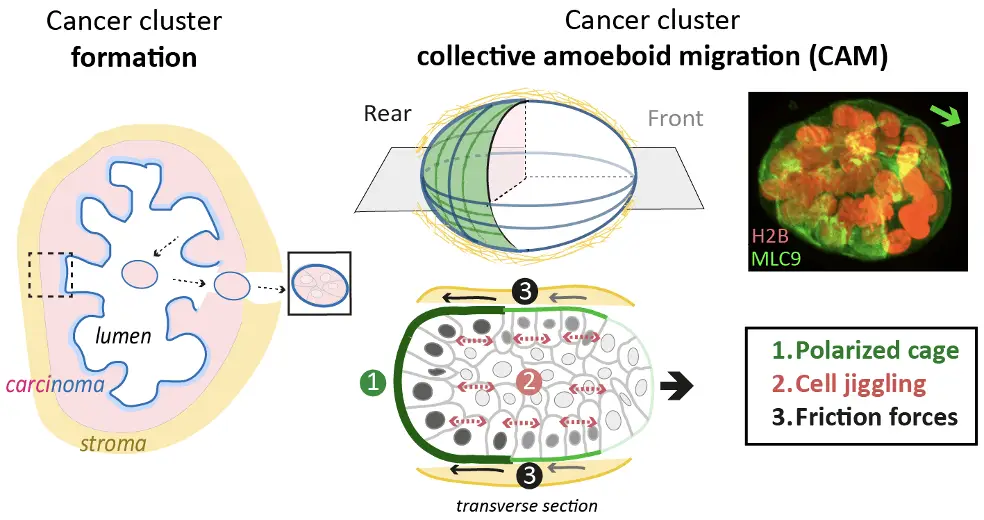
Déterminants moléculaires et cellulaires de la CAM
Polarité antéro-postérieure des amas tumoraux
Comment les cellules individuelles coordonnent-elles leur réponse aux signaux de rupture de symétrie pour définir l'axe de polarité supracellulaire avant-arrière de l'amas tumoral ?
Hiérarchisation leader/suiveur
Les cellules situées à l'arrière et à l'avant des groupes sont-elles réellement les suiveuses et les meneuses, respectivement ? Quelles sont leurs fonctions ? Sont-elles prédéterminées pour remplir leur fonction ?
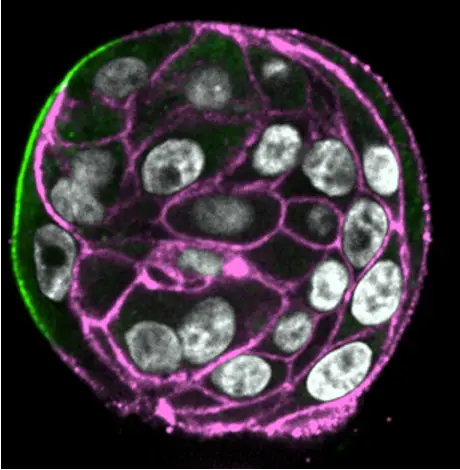
Biomécanique des amas de cellules tumorales lors de la CAM
État solide/liquide des amas tumoraux
La CAM est alimentée par le mouvement polarisé de cellules immobilisées à l'état solide (état vitreux, sans échanges avec les cellules voisines). Nous émettons l'hypothèse que, contrairement aux systèmes adhérents, la fluidification tissulaire inhibera la migration collective des amas subissant la CAM dans des environnements confinés non adhésifs. Nous avons établi une collaboration avec Giorgio Scita (IFOM, Italie) pour aborder ce domaine d'investigation.
Mécano-réponse des amas tumoraux aux forces externes (compression, cisaillement) lors de la dissémination
Comment les cellules individuelles au sein de l'amas coordonnent-elles leur réponse aux forces appliquées pour induire le mouvement de l'amas ? Quelles voies moléculaires sont impliquées ? En collaboration avec les laboratoires de Jacky Goetz et Gregory Giannone, Le programme METAGREG combinera des approches biophysiques et diverses approches d'invasion, allant de la microfluidique aux modèles animaux (danio zébré, souris).
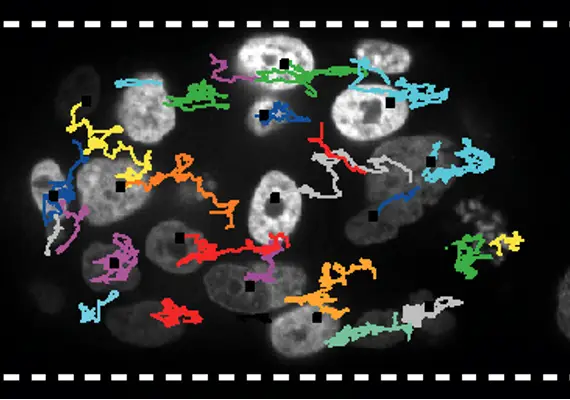
Plasticité de l'invasion collective du cancer
Déterminer le mécanisme cellulaire autonome et non autonome de la plasticité collective.
Les données préliminaires ont montré que, comme les cellules individuelles, les amas sont plastiques et alternent entre un mode de migration collective basé sur la traction et un mode basé sur la propulsion.
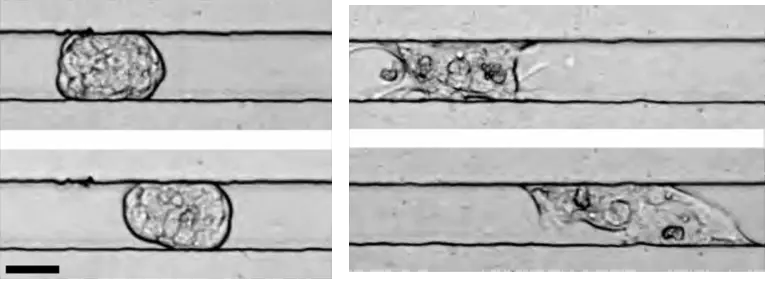
CAM et dissémination métastatique
À l'aide de modèles ex vivo et in vivo, nous étudierons si la CAM est plus efficace pour propager le cancer que la migration collective basée sur la traction.