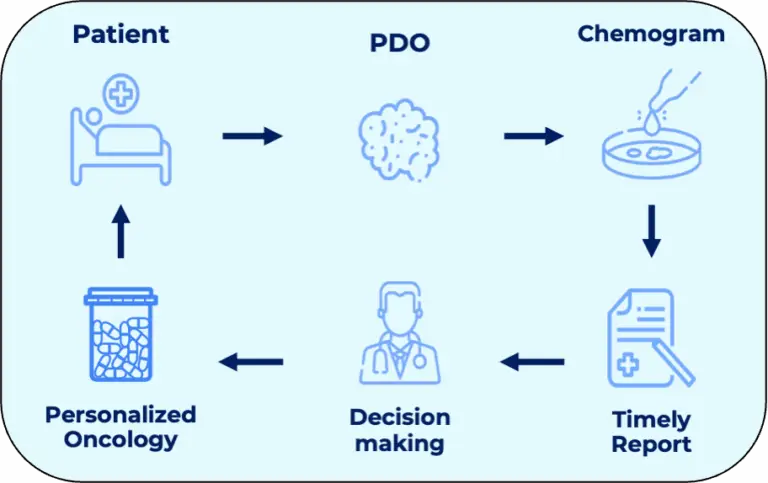
Patient-Derived Organoids for functional precision medicine strategies
Despite major advances, precision medicine based on genomics and targeted therapies currently benefits only 7-10% of patients. Our project aims to complement these molecular approaches with functional personalized medicine (FPM) approaches, based on the objective response of an ex-vivo drug test performed on patient-derived organoids (PDOs) (=TUMOR AVATAR). Performed in a one-patient one-test basis, this will identify effective drugs against the tumor, regardless of its molecular profile or anatomical site, as well as model and anticipate treatment resistance. Since their discovery 10 years ago, organoids have emerged as powerful tools but are not yet exploited in the clinic because of the lack of large prospective cohorts addressing the clinical validity and utility of this approach for solid cancers. At the interface between INSERM and Gustave Roussy hospital, the Jaulin lab provides the ideal ecosystem to address this challenge.
Our goal is to develop 3 tests that will be implemented in the clinic and contribute to generate new knowledge from patient tumor specimens:
- The chemogram will test the sensitivity of each organoid to a wide range of drugs (chemotherapies, targeted therapies) enabling to choose the best treatment and avoid the unnecessary toxicities of inefficient treatments.
- The immunogram will predict the response of each patient’s immuno-oncology treatments, from immune check-point blockers to adoptive cell therapies (CAR-T).
- The invagram will predict the aggressiveness (proliferation x invasion) of cancers to enable the personalized monitoring of patients at risk of relapse
RHU ORGANOMIC
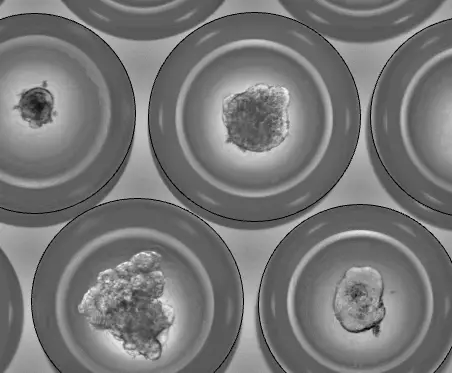
Based on our seminal work on PDOs (Roy et al. 2017), this “Bench-to-bedside” research program has been endorsed and granted by a RHU grant (ORGANOMIC). Our consortium of academic and industrial partners aims to develop in vitro diagnostic tools based on PDOs. It will combine cutting-edge technologies such as artificial intelligence, mathematical modeling, microscopy-based approaches, and microengineering to generate “tumor-on-chips” intended to standardize and automate on a large-scale tests performed on organoids derived from each patient.
Preclinical studies: ORGANOPREDICT
“Clinical Implementation of a Personalized Medicine Strategy Based on PDOs”.
For FPM strategies to patients, we need
- Scalability: grow PDO for most patients from tiny needle biopsies (take-on rate),
- Temporality: in order not to leave patients without treatment, we had to be able to establish, amplify and drug-test PDOs as quickly as possible (turnaround time),
- Predictability: We must determine the sensitivity and specificity or the chemograms to evaluate their predictive value. Our two pilot studies on colorectal & pancreatic cancers including respectively 40 and 81 patients proved very similar results. (see publications: Cartry et al, 2023 J Clin Exp res; Boileve et al, Gastroenterology, 2024)
We generated organoids for 63% of patients and produced the chemogram report in 6.3 weeks on average for a panel of 25 anticancer drugs. Our assay harbors a sensitivity of 93% and specificity of 84%. Importantly, for 81% of patients, we identified at least one effective molecule and three effective molecules on average in our cohort. These results and the improvement of the technology have proved that despite constraints, organoid technology could be tested in interventional clinical trial, provided that the biopsy is performed at the beginning of the last treatment line.

Clinical studies: ORGANOTREAT

We opened this clinical trial in 2022. Covering all solid tumors, it is the first national clinical trial of personalized medicine based on PDOs and one of the first internationally, allowing us to take a strategic position in the field. This is an interventional multicenter academic trial promoted by Gustave Roussy aiming to determine the feasibility and efficacy of functional personalized medicine based on organoids.
The first segment of this trial is a phase I/II titled ORGANOTREAT-01, which aims to test the feasibility and effectiveness of a personalized drug test conducted from organoids derived from 60 patients with colorectal cancer which has just been completed. Our primary objective is already positive as we have generated chemograms for 81% of patients and perform the test in 6.1 weeks.
- The proportion of patients actually treated in accordance with CTB recommendations and,
- Their tumor response to treatment based on their chemogram (measuring the “Growth Modulatory Index or GMI (corresponding to the ratio of progression-free survivals with the previous treatment line (PFS2/PFS1)), these data are currently being collected.